While some errors are unforeseen, many can be prevented with better planning, coordination, communication and transparency in launch planning and implementation. In our article for PME, we make the case for why pharma companies need a launch execution platform that makes launch excellence the norm, builds the corporate launch memory, and streamlines efforts to save time for everyone involved in product launches.

The importance of a pharma product’s launch to its success cannot be overstated. A recent analysis by Deloitte showed that most new drugs continue with the revenue trajectory set at launch – around 70 percent of products that miss expectations at launch continue doing so in subsequent years, and around 80 percent of products that meet or beat expectations continue to do so afterward.
In other words, you’ve got one shot to get it absolutely right.
And with ever-increasing development costs and in an ever-more competitive landscape, the need to get it right is greater than ever. But it’s more complex these days, too. Whether it’s more specialty products, evolving regulatory frameworks, increasingly diverse territories, increasing payer influence, or the need for multi-channel communication, the complexity of a global product launch is only increasing. COVID-19 – and the subsequent demise of traditional face-to-face sales representative time – has only added to the list of challenges.
|
You can find the entire May 2021 edition of PME here.
|
Many companies are struggling to get it right. According to a recent report by L.E.K. Consulting, half of drugs rolled out since 2004 didn't live up to their sales forecasts. And according to IQVIA, fewer than 10% of launches are categorized as ‘Excellent’.
So why does it go wrong so often?
There are many potential reasons, including a lack of engagement with key medical and payer stakeholders, an inadequate pricing and market access strategy, or an overstated valuation by the company. But while some errors are unforeseen, many can be prevented with better planning, coordination, communication and transparency in launch planning and implementation.
Building a solid launch pad: launch excellence frameworks
Just as rocket launches need a dependable launchpad, so pharma company launches fly better from a solid foundation, in the shape of a best practice launch excellence framework. After all, why spend resources on relearning lessons that others in your company have already learned?
Elements of your launch excellence framework may include, for example, best practices around cross-team structures, analytics to understand the market, strategy to understand the critical success factors and key performance indicators, as well as approaches to stakeholder engagement and local market roll-out.
The various elements of a framework can broadly be divided into three core buckets that companies must try to shape: the market, the product, and the organization (see image below).
Launch Excellence Framework
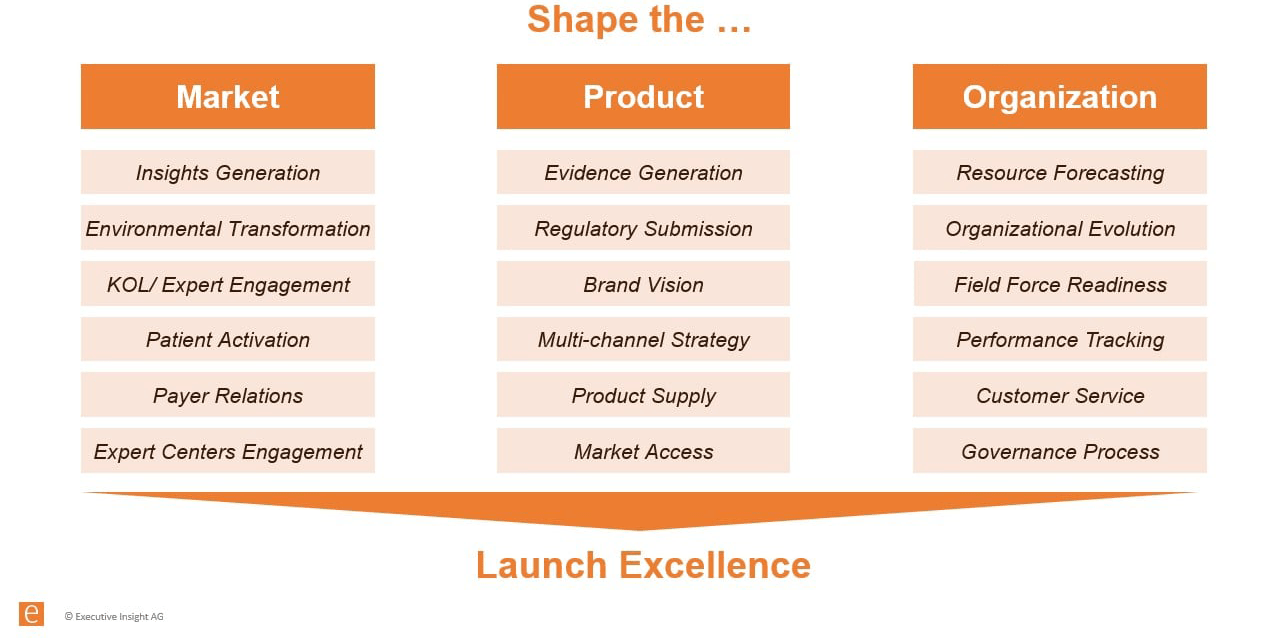 Source: Executive Insight
Source: Executive Insight
Having a ‘template’ launch excellence framework embedded across the business avoids duplication of effort and resources. It enables launch teams to focus on tailoring this template to the specifics of their product and market, rather than reinventing the wheel by creating their own launch framework or launch planning tools.
From an affiliate perspective, the launch excellence framework should provide a common starting point for all of them, which reinforces the company’s strategic drivers. At the same time, it allows enough flexibility for each country to tailor its launch activity plan around the specific needs of their market. Close collaboration and co-creation with affiliates (potentially followed by training) is essential to ensure understanding and buy-in.
So far, so good. But here’s where it so often falls down. Unless the framework is truly embedded across the organization, in plain sight of all team members, and can be as flexible and adaptable as required, things can start to go wrong. Miscommunication, misunderstandings, deadlines missed, timelines slipping, duplication of effort… and once these things start happening, they tend to snowball.
'A good launch execution platform standardises excellence, builds the corporate launch memory, streamlines efforts and saves time for everyone involved'
Linking to mission control: digital launch execution platforms
This is where modern launch readiness software comes in. A dedicated launch execution platform can be used to optimize the impact of a launch excellence framework. It can ensure that the framework is fully embedded and systematically applied across the business, whilst allowing enough flexibility to address the particular needs of individual therapy areas, products and countries around each product launch. Companies can simply create a best practice launch execution template and allow individual product teams to tailor it each time, without starting from scratch.
Having the framework digitally embedded within the organization and at everyone’s fingertips– that means everyone involved in the launch, not just centralized launch project managers – will not only save time for each product team, but also ensure that they start off with a foundation that sets them on a path to a higher quality launch execution.
SmartLaunch™ – launch execution software from TRiBECA® Knowledge – gives companies full visibility across countries, workstreams and the whole launch portfolio. It aligns global and country launch plans, manages interdependencies, drives cross-functional collaboration, and automates and streamlines processes to save time.
Such platforms may have been viewed as a ‘nice to have’ option in the past. But in the era of COVID-19, with paths to launch cluttered with pandemic-related challenges, there has been a huge acceleration in the acceptance of transitioning to digital strategies to oversee launch campaigns. Both pharmaceutical launch teams and their customers are likely to become significantly more acclimatized to digital interaction in the coming months and years.
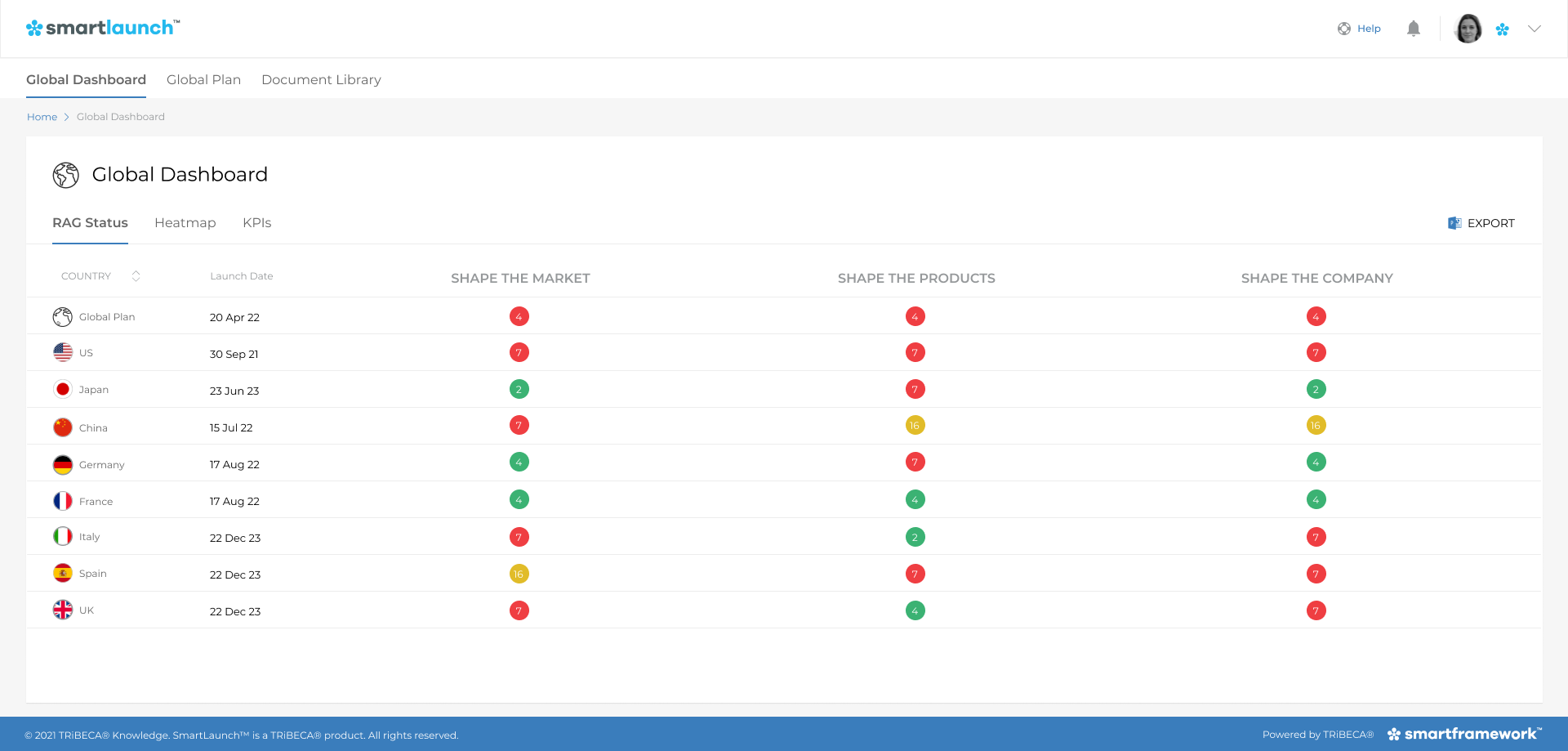
One of the greatest benefits these platforms bring is transparency. For launch teams stretched across geographies and time zones, the real-time dashboards and reports ensure everyone is fully aware of what will be delivered when and by whom, avoiding duplication of efforts. Activity plans can be tracked continuously, and in real-time, with an early warning system for risks and potential delays (see Image 2).
Platforms like SmartLaunch™ not only help to facilitate transparency and manage interactions between global and local activities. They can also automate communications, for example, to notify the relevant people in the country launch teams when there is a delay to a global deliverable.
The SmartLaunch™ traffic light dashboard
Ensuring a hitch-free ride: maximizing automation and digitalization
Because after all, some delays or hitches are inevitable. Unexpected events – whether new evidence emerging from clinical trials, regulatory approval letters or HTA assessments for example – should be expected. Activity planning needs to be agile to cope with new realities and changing deadlines.
The visibility offered by platforms like SmartLaunch™ means that any such events are communicated immediately to all parties. This is particularly pertinent in the COVID-19 era, when collaboration is made even more challenging by remote working. The dashboard enables all team members to get up to speed simultaneously and facilitates collaboration around activity planning, tracking milestones, tracking decisions and monitoring risks.
Automated interdependencies are also built into the platform, so that the knock-on impact of any new events or changing deadlines is automatically updated downstream. For example, SmartLaunch™ will flag if you are trying to move a date which has an impact on the launch date in a country. Furthermore, the platform will allow timelines to be adjusted automatically if the timelines for upstream dependencies (predecessor tasks) change or indeed if the launch date changes. This planning agility, with dashboards updated in real-time, is much needed in today’s fast-paced launch environment.
SmartLaunch™ also automatically chases people, reaching out when someone’s attention is needed, relieving the administrative burden and allowing more focus on strategic priorities.
‘Around 70% of products that miss expectations at launch continue doing so in subsequent years, and around 80% of products that meet or beat expectations continue to do so afterward'
Bringing it home again: making launch excellence standard
Elon Musk recently lauded reusable rockets as the “holy grail” of space travel. The same philosophy can be applied to product launches – if you’ve done it all perfectly one time, why not reuse as much as you can the next time?
All biopharma companies recognize the vital importance of the launch to a product’s success. It’s likely that most will have executed standalone launches in their history that could be considered best practice. But why not make this systematic, standard practice?
A good launch execution platform standardizes excellence, builds the corporate launch memory, streamlines efforts and saves time for everyone involved, whilst giving countries the visibility they need for their decision making. In an era when companies are increasingly overseeing a steady stream of launches for a series of sub-indications for a molecule, rather than just a one-off event, this need increases even further.
For product teams, a cutting-edge launch execution platform can achieve significant time efficiencies, whilst ensuring that the team begins on a path to a high-quality launch execution. For the company, it means that they can reinforce and consistently embed the launch framework across the business, making sure that all launch plans contain the key activities that should be mandatory for strategic, regulatory, commercial or legal reasons.
Launch execution platforms like SmartLaunch™ allow biopharma companies to apply the same rigor and failsafe approach to launch execution as they already do with their R&D project management. Without platforms like this, biopharma companies run the risk of stumbling at the last step of a long and costly journey to launch, and limiting the number of patients who will benefit from their innovation.
It’s time to remove that risk, launch with confidence and shoot for the moon.
The Authors
Aleksandar Ruzicic is Partner; Paul Gardiner is Director; Tom Hartwig is Consultant at specialist healthcare consultancy Executive Insight. André Moa is CEO of pharma-tech company TRiBECA® Knowledge.







 Andre Moa
Andre Moa
 19 May 2021
19 May 2021
 10 minute read
10 minute read